Detecting water voles with environmental DNA surveys
Background
The water vole (Arvicola amphibius) is the UK’s most rapidly declining mammal due to a combination of habitat degradation and depredation by the non-native American mink (Neovison vison). Detection and monitoring of water vole populations is important for conservation schemes and is required for planning authorities wishing to build on or alter habitat that is suitable for water voles. Traditional water vole surveys involve looking for visual signs of their presence including tracks, burrows, latrines, feeding stations and live individuals. However, water voles are very elusive and are less active during the winter, meaning there is a need to develop more reliable and practical tools for determining presence or absence.
One solution could come in the form of environmental DNA (eDNA); DNA that can be extracted from abiotic samples such as soil, sediment and water, and which arises from the depositing of faeces, urine, saliva and epidermal cells into the environment (Taberlet et al. 2012). So, if we can detect water vole DNA in water samples, we can infer their presence (or absence if there’s no DNA) at a site without needing to see any physical signs of their presence.

My research
For one of my master’s projects at Imperial College London, I worked on designing and testing an eDNA survey for water voles. First I designed species-specific primers and a hydrolysis probe for use in qPCR, which I tested against water vole DNA. I then tested the survey on water samples collected and filtered from the water vole enclosure at the British Wildlife Centre, where there would definitely be water vole DNA. Once I had confirmed that my survey methods could detect water vole DNA in water samples, I had to test it in a more natural setting. I therefore conducted eDNA surveys alongside traditional surveys at four sites in South East England; three known to host water voles and one where water voles were known to be absent. I successfully detected water vole DNA at two sites where water voles were expected to be present, despite finding physical signs of them at only one of these sites. As expected, I did not detect water vole DNA at the site where water voles were absent.

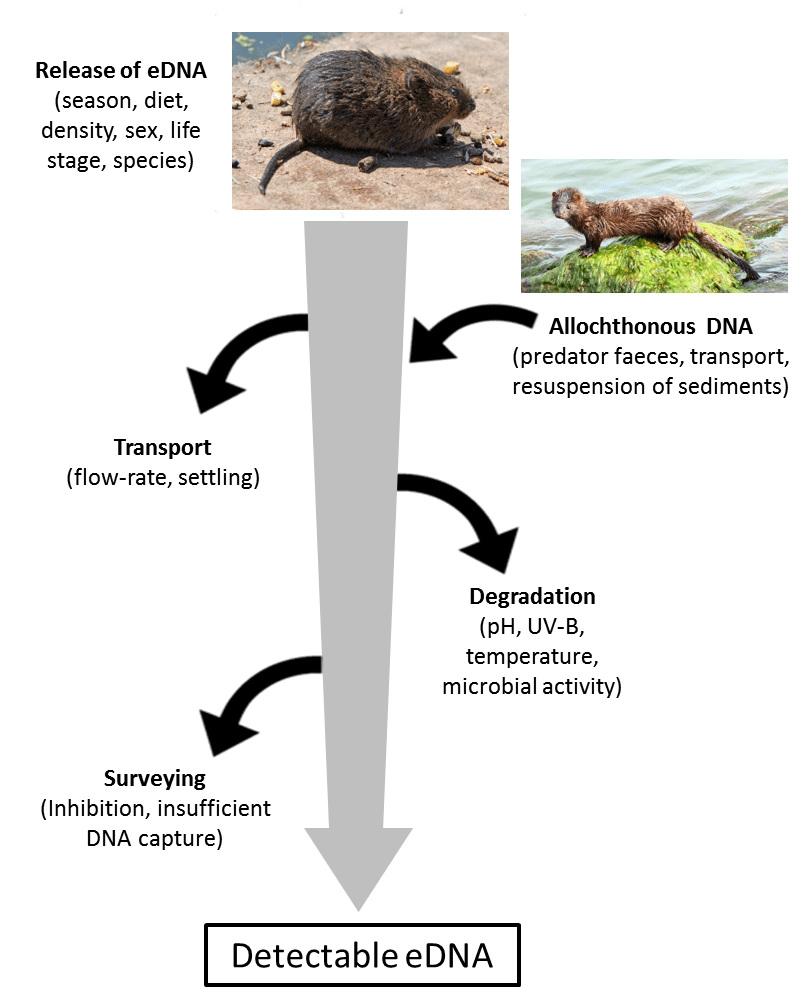
With only having five months to design and test the survey then write up my dissertation, there is still much to research before eDNA surveys for water voles are commonplace among ecologists and conservationists. In particular there are many environmental factors (e.g. flow rate, pH and temperature of the water (Figure 1)) that could affect if water vole DNA is detectable in water samples or not. These factors require considerable more research before the results of such surveys can be reliably interpreted. The Savolainen lab at Imperial College London are continuing with this research and we hope to publish our findings in due course.